Introduction
The idea of ‘clean’ water has evolved over the years as we further our understanding of human impact on the environment, and the subsequent impact of a polluted environment on our health. The 20th century brought a wealth of technological advancements that improved our ability to mitigate our impact on environmental systems and protect the public from the health issues related to unsafe drinking water. Water and wastewater treatment have become critical aspects of our society’s ability to coexist with Earth’s natural systems. As industrial activity and population continue to grow in the 21st century, continuing improvement of water treatment technology is of vital importance.
COD vs. TOC: Chemistry of Water Treatment
There are a wide variety of chemical parameters, both organic and inorganic, that interact through complex reactions to perform water and wastewater treatment processes. A full treatment system is composed of a series of these processes, with the intention of eliminating all unwanted compounds from the influent stream. These systems are designed to provide stable and consistent removal of organic and inorganic matter, provided that the parameters that facilitate the reactions are maintained at proper levels. Therefore, operation of treatment plants requires extensive means of monitoring the essential water quality parameters throughout the plant. A brief, non-exhaustive list of common parameters is provided below:
Physical parameters:
- Total Suspended Solids (TSS)
- Volatile Suspended Solids (VSS)
- Electrical Conductivity (EC)
- Temperature
Inorganic chemical parameters:
- Ammonia (NH3)
- Nitrate (NO3)
- pH
- Alkalinity
Organic chemical parameters:
- 5-day Biochemical Oxygen Demand (BOD5)
- Chemical Oxygen Demand (COD)
- Total Organic Carbon (TOC)
- Dissolved Organic Carbon (DOC)
Requirements for measuring each parameter varies based on what is being treated (water/wastewater), volume being treated (influent flow rate), and specific treatment processes in use. Of the three groups mentioned, organic parameters tend to be the most important to operators, as it provides them with an actual measurement of influent organic load, organic removal from each process, and verification that effluent is within compliance. Many biological treatment processes also depend on a consistent organic load, and monitoring is essential to avoid upsets to biological activity.
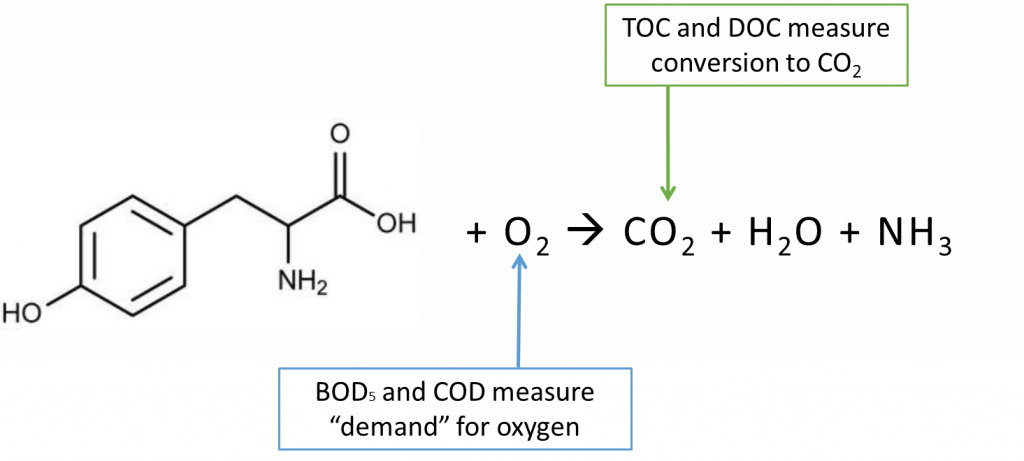
From a chemistry perspective, the mentioned organic parameters each provide a slightly different view of the organic compounds within water or wastewater. BOD5 is a test involving a 5-day incubation of sample with microbes, measuring the depletion of oxygen directly resulting from the biological activity facilitated by the organic matter in the sample. Samples containing higher concentrations of organic matter provide more ‘food’ for microbes, resulting in greater microbiological activity, leading to greater oxygen depletion. COD is a similar measure of organic matter and oxygen demand; however, the method of oxygen depletion is through chemical oxidation reactions as opposed to biological activity. TOC and DOC provide a measure of the total amount of carbon in a sample through determination of CO2 generation from an oxidation reaction. The graphic below summarizes the chemistry of these parameters:
From the chemistry, it can be seen that these parameters complement each other in the sense that they capture two separate, but related aspects of the organics in the sample. Oxygen demand, BOD5 and COD can give an indication as to the oxidizability or reactivity of the organics. TOC and DOC quantify the amount of carbon in the organics. By comparing these two aspects, insight can be gained on the overall ‘oxidation state’ of the organics.
Drinking Water
One of the most critical challenges faced by water utilities is quantification of organic load on their treatment system. The removal of natural organic matter (NOM) is crucial for maintaining stability of treated water within the distribution system. Inadequate removal of NOM can lead to taste/odour challenges, deterioration of pathogen log removal/inactivation capabilities, and formation of carcinogenic disinfection by-products (DBPs). In addition, water utilities that do not properly address NOM removal can see challenges with:
- Over-dosing coagulant during periods of low NOM
- Under-dosing coagulant during periods of high NOM
- Poor floc formation or settling
- Shorter filter run times
- More frequent backwashes
- Increased sludge production
- Reduced hydraulic capacity
- Membrane fouling and associated impacts
- Reduced effectiveness of ion-exchange/adsorption processes
Health Canada has recognized this knowledge gap, and to help utilities understand and address the challenge, they will release in Fall 2018 a guidance document focused specifically on Natural Organic Matter in Drinking Water. Within this document, the topic of quantifying NOM is explored thoroughly, and the common surrogate measures are compared. While optical measures such as color, UV254 and SUVA are widely used due to low price and simplicity, these are surrogate measures and must be calibrated on a matrix specific basis. Comparison to other utilities, therefore, is quite difficult. TOC and DOC provide an empirical quantification of the carbon in NOM, however they give no indication as to the reactive state of the NOM compounds.
COD provides an insight into oxidizability (or reactivity) of the NOM in a water sample, however common methods for determining COD involve hazardous reagents and waste, and a 3-hour test time, which make the test unsuitable for water utilities to perform on-site. In addition, the common method is not sensitive to lower concentrations often seen in source water for WTPs. Due to these shortcomings, COD has historically not been considered for source and treated water monitoring.
Photoelectrochemical Oxygen Demand (PeCOD) is a novel technique for determining COD that eliminates the hazardous chemicals and delivers a result within 5 minutes for water samples. A 2014 study by Dalhouise University highlights PeCOD as an excellent opportunity for drinking water utilities to address NOM removal with a green, easy-to-use method that can be directly implemented in the plant either in portable, benchtop, or online configurations. PeCOD has already proved invaluable to numerous utilities through its ability to monitor for DBP formation, optimize coagulation processes, and identify deficiency in a treatment process quicker than other common measures. An ASTM International method for PeCOD, D8084, was released in 2017 and confirmed the applicability of PeCOD to drinking water treatment, showing an MDL for the test of 0.7 mg/L.
Waste Water
Wastewater treatment plants are highly dependent on biological processes to break down the wide variety of organics stemming from both sanitary sewage and industrial process effluents. Upon initial plant design, all aspects of the expected influent waste are taken into account, and ultimately determine the appropriate combination and configuration of treatment processes. The expected BOD, COD, and TOC are crucial in this determination, as they are directly related to the microbes’ ability to thrive in their appropriate treatment process. If the microbes are not supplied with enough organics (or ‘food’), a die-off will occur impacting overall treatment performance. If the microbes are supplied with too much organic matter, they will propagate beyond the intended design parameters and cause numerous plant challenges such as clogging, stagnation, odor events, and decreased effluent quality.
After a treatment plant becomes operational, one of the greatest challenges faced is keeping up with constantly increasing wastewater loads in growing areas. Plant design typically includes a factor accounting for expected growth in the service area, however it can be quite difficult to estimate industrial expansion and subsequent increases in industrial wastewater. In the case of process effluents, the challenge is not only in determining the increase in volume, but also the increase in organic load and oxygen demand. As industrial activity in a service area develops, it is in the hands of the treatment plant and municipality to determine the changes in wastewater volume/quality and make adjustments to the treatment processes to account for it.
The ability of a wastewater utility to keep up with changing/increasing load hinges on their ability to detect the changes. With respect to organic properties, the ability to detect changes is highly dependent on the method of quantifying organic load. Dr. Andrea F. Gullà of Thermo Fisher Scientific addressed this in a recent article on the topic of Robust Water Management, in which he investigates some of the pressing challenges seen worldwide with respect to changing wastewater loads and true quantification of organic load. On the wastewater side, focus is given to the challenge presented by pharmaceutical products and drugs in the wastewater stream. These can be very difficult to detect and quantify due to the complex structure of the organic molecules, and they have a greater chance of passing through treatment without getting broken down. For this reason, pharmaceutical manufacturers are required to treat their wastewater effluent before discharging to the sewer system, however utilities are still posed with the challenge of detecting and treating pharmaceuticals and drugs contributed by sanitary sewage.
TOC is a common parameter that utilities use to monitor for changes in organic load. However, studies have shown that this test is not sufficient for determining the full impact of complex organics on treatment. Organics with similar amount of carbon may have vastly differing molecular structures; therefore COD provides insight into the oxygen demand, and reactivity, of the wastewater load. While COD is a common parameter for wastewater utilities to monitor within the plant, the 3-hour time requirement for the standard dichromate COD test makes it unsuitable for use as an influent and effluent monitor. PeCOD is a new method of COD determination that solves this challenge, providing a COD result in less than 12 minutes. This allows the utilities to be proactive and fine-tune their treatment processes based on the actual organic load at any given time. In addition, the oxidation potential of the PeCOD chemistry is double that of the dichromate method, which indicates that it is more suitable for detecting and quantifying oxygen demand of complex organic molecules.
Conclusion
As we move forward in the 21st century, utilities and industries are under constant pressure to improve their treatment processes. Safeguarding human health and the environment is of critical importance, and as population and industry expand so too must our ability to monitor and treat the water and wastewater we produce. Treatment plants, both municipal and industrial, rely on their monitoring tools to provide optimal treatment and benefit greatly from having as many of these tools available as possible. PeCOD is the tool which can deliver the COD results quickly, effectively, and with the greatest ability to detect changes. It is simple to operate in any environment, 24/7, and a low cost for investment.









Is there any standard limit of TOC for industrial effluent discharge or for drinking water like other paramiters such as TDS, TSS, COD etc. ?
TOC is sometimes industrial compliance parameter for Petrochemical industries and some Chemical manufacturers. In all other cases, if TOC is being used, it is to correlate to COD and/or BOD. Therefore, it is better to use actual COD method from an analytical point of view. MANTECH’s PeCOD is a rapid, green, and easy-to-use analyzer for empirical COD, and is ideal for industries that are using TOC results for internal purposes (process optimization, effluent monitoring, etc).
TOC is commonly used as a guidance for drinking water, however, Specific UV Absorbance (SUVA) is often regarded as a better parameter to monitor for Natural Organic Matter (NOM) in drinking water applications. This is a combination of TOC and UV Absorbance at 254nm (UV254), providing insight to the quality of the organic matter rather than just quanitifying carbon via TOC. MANTECH’s PeCOD is shown to have a strong correlation to SUVA, and has been described as a “2 for 1 SUVA”, one instrument and one sample to measure NOM reactivity in minutes. You can read a brief by Dalhousie University on this topic here: https://mantech-inc.com/blog/dalhousie-cwrs-application-brief-a-comparison-of-pecod-vs-specific-uv-absorbance-for-monitoring-nom-reactivity/