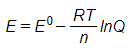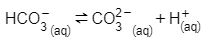Alkalinity, pH , and conductivity are just a few of many parameters that are highly affected by temperature. In order to ensure accurate measurements, it is critical to maintain the temperature of the measurement by either a) compensation via mathematical correction, or b) keeping the sample to a constant reference temperature.
pH
The slope calculated by a pH electrode follows accordingly with the Nernst Equation:
Where, E is the output from the electrode (mV), E0 is the standard potential, R is the gas constant (8.314 J•mol-1•K-1), T is the temperature (K), n is the number of electrons, and Q is the reaction quotient. Clearly, depending on the temperature of the sample, the mV reading from the electrode will change, and therefore, must be accounted for.
Furthermore, based on Le Chatelier’s Principle, by increasing the temperature of a sample, the molecules ability to release hydrogen ions (H+) increases due to their increased molecular vibrations. As a result, the pH will effectively decrease as more H+ dissociates.
Alkalinity
On the other hand, alkalinity measures the ability of a solution to neutralize acid, therefore the dissociation equation is as follows:
Increasing the temperature will both increase the amount of carbonate that is available and increase the H+ concentration. As a result, the buffering capacity will increase, and the pH will decrease.
Conductivity
As stated above, increasing temperature will increase the mobility of ions in solutions as they gain kinetic energy. Since conductivity measures the capacity of a solution to conduct electric current, ions will be able to move to the plate of opposite charge more easily. In turn, the conductivity reading will be increased.
How can MANTECH assist your company?
MANTECH offers both automated temperature compensation and temperature-controlled methods with all MT-Series models for multi-parameter automated titration analysis. Temperature compensation is available via manual temperature entry with the MT-10 or by thermistor with the MT-30 and MT-100.
Moreover, MANTECH offers a water bath to regulate the sample temperature. This method is available both with and without a top rack to retain sample tubes and cups that may float in water, if needed. Another method of temperature control available is to use a water jacketed analysis vessel. The water jacketed vessel is used to control the temperature of the sample by constantly circulating temperature-controlled water around the vessel. The temperature of the sample is maintained constant by heat transfer through the glass vessel. MANTECH offers several volumes for the vessel including 35 mL, 85 mL, and 145 mL for the TitraSip™ module (MT-100 systems). For example, for automated alklainity at a constant 25 degrees C, MANTECH recommends the 35 mL vessel.
Author:
Laura Martin, Quality Control and Research Chemist