Fluoride by Standard Addition
Method Abstract #48
Scope and Application
This method determines the concentration of fluoride ions in a sample and helps overcome sample matrix difficulties.
Method Summary
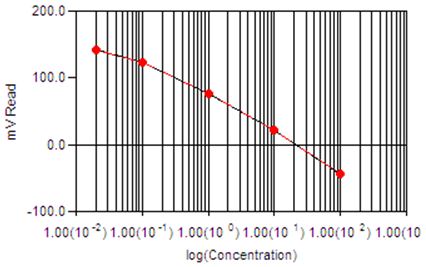
The concentration of fluoride is determined by a fluoride ion selective electrode (ISE). An initial reading is taken, following which a known amount of a higher concentration standard is added to the sample, via buret. A second measurement is then taken and based on the millivolt (mV) difference the concentration of the initial sample can be calculated. Both the standards and the samples are pre-treated with TISAB (Total Ionic Strength Adjuster) to ensure that the background response from each sample is similar.
Sample Calibration Curve
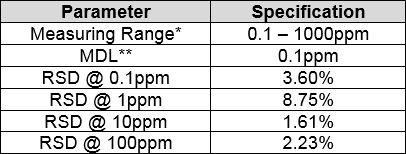
Method Performance
*Data for this measuring range was obtained using laboratory prepared standards formulated from sodium fluoride. The measuring range may be increased by using auto-dilution.
**The Method Detection Limit (MDL) was determined based on data obtaining a coefficient of variance better than 30%. Results may differ depending on laboratory practices and sample matrix.